 
The difference in behavior might have been because magnesium was in a solid form and the calcium was dehydrated and powdered, which appeared to mix with the water easily and dissolve. Wash test tubes.ġ.) What might be a reason for the difference in behavior between magnesium and calcium placed in water? Procedure - Part 1: Trends in Properties Within GroupsĤ.) Fill a beaker with tap water and get a disposable pipette.ĥ.) Bring test tubes to Charlene to get a sample of magnesium and a sample of calcium.Ħ.) Use pipette to cover sample with water.ħ.) Observe reaction carefully and enter data in table.Ĩ.) Dump sample into hand in sink, rinse sample, and place in corresponding labeled beaker. Gallium is in Group 3 and the 3rd period. The alkali metals are the first group on the periodic table and the alkali earth metals are the second group.Ĥ.) What element is in Group 3 and the 3rd Period? The metals are located to the left of the metalloid boundary.ģ.) What are the names of two metal families? 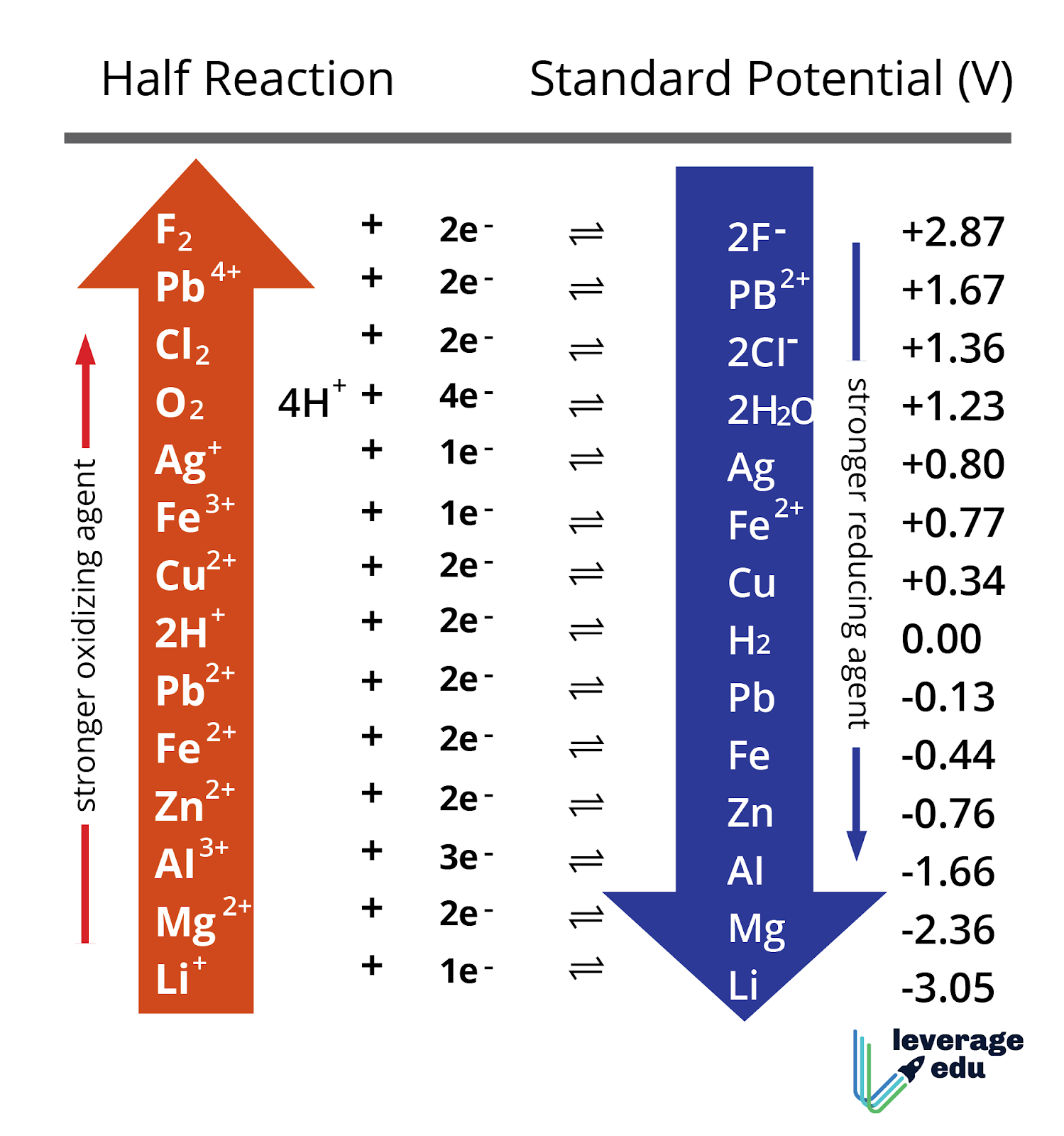
The groups of the periodic table that share a lot of similarities with all of the other elements in that group are called families.Ģ.) In general, where are the metals located on the periodic table? The periods of the periodic table are the horizontal rows. Groups of the periodic table are the vertical columns. To explore the reactivity trends of metals in groups and periods of the periodic table.ġ.) What are groups? What are periods? What are families?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
